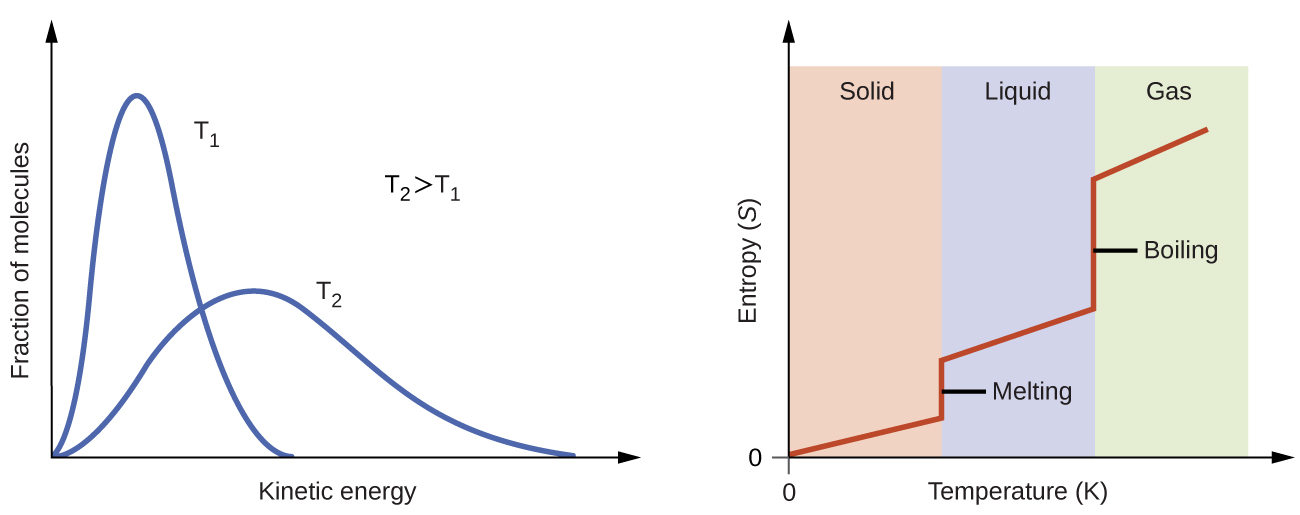
The Third Law of Thermodynamics is usually stated as a definition: the entropy of a perfect crystal of an element at the absolute zero of temperature is zero. You have probably heard the phrase “the ever expanding Universe”, although this phrase is often taken to mean our ever increasing knowledge of our Universe, it also is quite true that our Universe is expanding and this effect is entropic in nature. Coffee in cream and gas expansion and perfume in air are ‘how widely' processes where the initial energy of the molecules stay the same but the volume occupied by the molecules increases. You see now how hot pans cooling and chemical reactions belong to the ‘how much' category where energy is being transferred. This is reflected in the idea that a spontaneous change always results in the dispersal (spreading out) of energy (and also matter).

Entropy change measures the dispersal of energy: how much energy is spread out in a particular process, or how widely spread out it becomes (at a specific temperature). The Second Law of Thermodynamics states that for a reaction to be spontaneous, there must be an increase in entropy.Įntropy is often defined as a measure of the disorder of a system, this is not a very accurate definition. The best indicator of spontaneity in a reaction is the change in Entropy (S or DS). The application of spontaneity with respect to equilibrium is as follows:Ī reaction will never spontaneously move away from equilibrium and will always move spontaneously towards equilibrium.Īlthough spontaneous reactions are often exothermic, this is not a criteria for spontaneity.
A non-spontaneous process needs a continual input of energy. \(\text.A spontaneous process is one that, once started, continues on its own without input of energy. Note that the idea of a reversible process is a formalism required to support the development of various thermodynamic concepts no real processes are truly reversible, rather they are classified as irreversible. The term reversible process refers to a process that takes place at such a slow rate that it is always at equilibrium and its direction can be changed (it can be “reversed”) by an infinitesimally small change is some condition. This new property was expressed as the ratio of the reversible heat ( q rev) and the kelvin temperature ( T). In a later review of Carnot’s findings, Rudolf Clausius introduced a new thermodynamic property that relates the spontaneous heat flow accompanying a process to the temperature at which the process takes place. In 1824, at the age of 28, Nicolas Léonard Sadi Carnot ( ) published the results of an extensive study regarding the efficiency of steam heat engines. Predict the sign of the entropy change for chemical and physical processes.Explain the relationship between entropy and the number of microstates.Spectroscopic and Magnetic Properties of Coordination CompoundsĪldehydes, Ketones, Carboxylic Acids, and Estersīy the end of this section, you will be able to: Occurrence, Preparation, and Properties of Transition Metals and Their CompoundsĬoordination Chemistry of Transition Metals Transition Metals and Coordination Chemistry Occurrence, Preparation, and Properties of the Noble Gases Occurrence, Preparation, and Properties of Halogens Occurrence, Preparation, and Properties of Sulfur Occurrence, Preparation, and Compounds of Oxygen Occurrence, Preparation, and Properties of Phosphorus Occurrence, Preparation, and Properties of Nitrogen Occurrence, Preparation, and Properties of Carbonates Occurrence, Preparation, and Compounds of Hydrogen Structure and General Properties of the Nonmetals Structure and General Properties of the Metalloids Occurrence and Preparation of the Representative Metals Representative Metals, Metalloids, and Nonmetals The Second and Third Laws of Thermodynamics Shifting Equilibria: Le Châtelier’s Principle Stoichiometry of Gaseous Substances, Mixtures, and Reactions Relating Pressure, Volume, Amount, and Temperature: The Ideal Gas Law Periodic Variations in Element Properties Mathematical Treatment of Measurement Resultsĭetermining Empirical and Molecular FormulasĮlectronic Structure and Periodic Properties of ElementsĮlectronic Structure of Atoms (Electron Configurations) Measurement Uncertainty, Accuracy, and Precision
